BOISE, Idaho — A group of healthcare professionals in Blaine County is hoping to change federal policy.
This comes after the federal government told them twice to stop testing for COVID-19 antibodies.
Cathy Swink is the director for the county's COVID-19 Response Group. She wants to get federal policy changed so they can test people for antibodies and release the results on site.
The response group was formed because when the coronavirus pandemic first hit Idaho, Blaine County was the epicenter. it was one of the hardest-hit areas in the entire country. The group is comprised of two doctors, two pharmacists and a registered nurse.
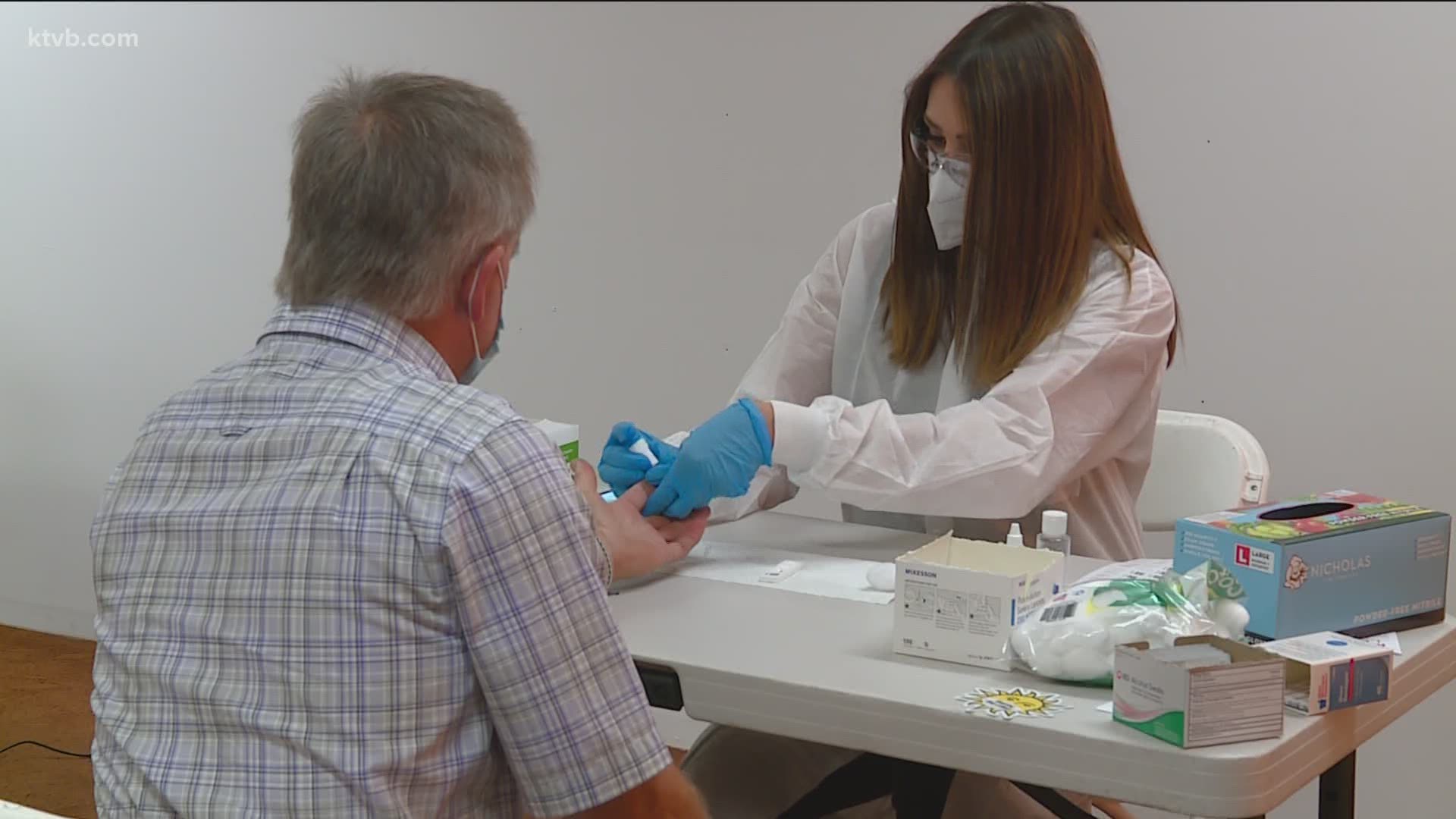
They’re currently conducting what is called a lateral flow assay study.
“That's really one of the main objects of our study was to find asymptomatic carriers,” Swink said. “Also, to look at the prevalence and also to show that these LFA antibody tests should be used in community settings such as pharmacies.”
The group first started testing individuals back in March.
“Everything was going smooth. We were very busy; we would fill up appointments within five seconds of releasing them online,” Swink said. “The demand was high, and the demand still is high.”
Their tests identify two different kinds of antibodies. One is IGG, which means the person has been exposed to COVID-19. The other is IGM, and this indicates the person has the active virus.
A difference between these tests and PCR tests, or ones that are used to find out if someone currently has the virus, is the turnaround time. Swink told KTVB that’s why this study is important.
“Especially today with PCR testing in Idaho being so delayed, some people are waiting up to 21 days for their results,” Swink said. “We’re able to provide results in 10 minutes and if they do show up for positive IGM antibodies then we can refer them to get a PCR.”
However, if someone does test positive for the IGG antibody, the group isn’t telling the patient that they’re immune, only that they’ve been exposed.
“No one can say you have IGG, you’re immune. Nobody can say that,” Swink said. “We don’t know how long antibodies are lasting for. We don’t know the level of antibodies you need.”
Swink said they re-tested some people in the last week who were tested in March as well. In March, those individuals showed the IGG antibody. The re-tests during the summer showed they no longer had those antibodies.
After they started the study, The Centers for Medicare and Medicaid Services, a division of the U.S. Department of Health and Human Services, told researchers that they needed to stop since they weren’t testing them in an approved lab. If they continued testing, CMS said it would issue a cease and desist letter, according to Swink.
“They require these tests to be done in a high complexity lab which is basically a hospital, an urgent care or a company like Quest or Interpath labs,” she said.
But testing in a CLIA certified laboratory isn't possible for them due to their location.
“So, we'd have to mail these tests to those labs,” Swink said, adding that mailing the tests would make them invalid.
“You have to read them within a certain timeframe, usually within ten minutes,” she said.
The group got an institutional review board to approve their study. Under FDA regulations, an IRB is a group of people that review and monitor medical research that involves humans.
“An IRB is basically to allow the use of these tests in a community setting, and to also show the numbers and educate our community if they have COVID or not or what to do next,” Swink said.
Even with the IRB, CMS issued a cease and desist letter in early August. The letter stated the group cannot keep testing without a CLIA certificate. CLIA stands for Certified Laboratory Improvement Amendments.
“In sum, in order to do COVID-19 testing you must be a CLIA-certified laboratory that meets applicable regulatory requirements,” the cease and desist letter reads. “Under CLIA, laboratories are prohibited from testing human specimens for the purpose of diagnosis, prevention, treatment, or health assessment without a valid CLIA certificate.”
The letter also states how to become a CLIA certified lab. In order to do this, labs must comply with certain requirements regarding accuracy, quality, and reliability as dictated by statute.
“The purpose of these requirements is to ensure that the information that patients or their health care providers receive from a clinical laboratory is accurate and reliable test results,” the letter stated.
Following receiving the letter, the Blaine County research group decided to continue testing but they would no longer release the results to the patients. However, the cease and desist letter informed the group they were required to cease all testing immediately.
“We’re not able to tell those asymptomatic people that have IGM to go get a PCR test,” Swink said. “It hurts the study and it hurts our community, more importantly.”
The letter sent to the group told them the test they use is categorized by default as a high complexity laboratory test. This is something Swink disagrees with.
“They should not have to go to a high sensitivity lab to be read,” she said. “They should be available at every pharmacy, every doctor’s office.”
The group will stop testing on Friday. If they don’t, they could face daily fines that would cost them more than $20,000.
The group is now working on changing the federal policy to allow them to continue testing.
“It is red tape, it is language," Swink said. "I guess it's language that should be changed."
KTVB emailed CMS a list of questions for this story. At the time of publication, the organization has not responded.
KTVB also reached out to three members of Idaho’s congressional delegation. Sen. Jim Risch and Rep. Mike Simpson didn’t send official responses back.
Sen. Mike Crapo’s office told KTVB that he’s aware of the group's concerns and is working with the delegation, Gov. Brad Little, and federal partners to secure increased testing capacity for Idahoans.
Facts not fear: More on coronavirus
See our latest updates in our YouTube playlist: